
Learning agenda on hepatitis B birth dose vaccine delivery in Africa: Insights and recommendations
With funding from Gavi, the Vaccine Alliance, PATH employed a mixed-methods approach to assess learning questions on the feasibility, acceptability, cost, market access, and impact of innovative strategies to improve the reach of timely hepatitis B birth dose for babies born both within and outside of facility settings.
Chronic hepatitis B affects an estimated 254 million people worldwide, with the greatest burden in low‑ and middle‑income countries, particularly in the World Health Organization (WHO) African Region. Most chronic infections begin in infancy through vertical transmission at birth, which carries a 90 percent risk of lifelong infection, or through early childhood infections, which pose a 30 percent risk. WHO recommends administration of the hepatitis B birth‑dose vaccine within 24 hours of birth, which can prevent up to 95 percent of vertical transmissions. Yet many countries struggle to achieve timely coverage, often where large proportions of births occur at home.
With support from Gavi, the Vaccine Alliance, PATH conducted a mixed‑methods assessment to examine the feasibility, acceptability, cost, and impact of strategies for timely birth‑dose vaccination to infants born both in and outside health facilities. The project explored delivery models, the role of community health systems, and stakeholder perspectives to identify effective approaches for increasing coverage.
The project was implemented in four African countries: Ethiopia, The Gambia, Nigeria, and Uganda. The Gambia and Nigeria are among the earliest adopters of the hepatitis B birth dose in sub-Saharan Africa, whereas Ethiopia and Uganda have recently introduced the vaccine. Learning summaries from each country are available in the Resources section below. These findings can guide countries introducing or strengthening efforts to implement the hepatitis B birth dose.
Methods
The study applied a mixed‑methods design to understand how countries can strengthen timely hepatitis B birth-dose delivery, both within and outside health facilities. It explored global and country‑specific evidence through a rapid scoping review of literature and a desk review of national policies and studies. These efforts informed the design of data collection tools and provided essential context for four in‑depth country case studies. The team also conducted extensive interviews and focus group discussions with diverse stakeholders—from national policymakers to frontline health workers, caregivers, and community members—to uncover the practical challenges, opportunities, and factors influencing birth-dose delivery.
The project also included market research examining vaccine presentation options and the feasibility of using controlled temperature chain strategies, while demand forecasting modeled the required number of doses across various scenarios for low‑ and middle‑income countries. Cost modeling assessed wastage and delivery costs for different vial sizes and delivery settings, and human‑centered design sprints generated prototypes for improving both in‑facility and out‑of‑facility birth-dose vaccine delivery. Results were analyzed within each country to answer the project learning questions and then synthesized across countries to identify common themes, key barriers, and promising strategies for improving hepatitis B birth-dose coverage.
Barriers and facilitators to timely hepatitis B birth dose delivery
The project identified key barriers and facilitators to timely hepatitis B birth-dose vaccination at the community and facility levels that were common across all four countries. Barriers and facilitators are organized by four stages of the immunization journey, based on a modified version of UNICEF’s Journey to Health and Immunization framework: knowledge, awareness, and belief; intent, preparation, cost, and effort; experience of care at point of service; and after service follow-up. Across the framework stages, common factors emerged that shape whether caregivers and providers act in ways that support timely birth-dose vaccination:
- Caregivers’ connection to the health system—particularly through community health workers (CHWs)—shapes pregnancy preparation, delivery decisions, and timely identification and follow-up of home births.
- Overstretched health workers and weak community–facility communication limit caregivers’ access to early counseling; delivery and postnatal support; and follow-up care.
- Mothers’ experiences at facilities influence their decisions on delivery location and postnatal care; lack of respectful care discourages facility births and can deter women from seeking newborn vaccination after home delivery.
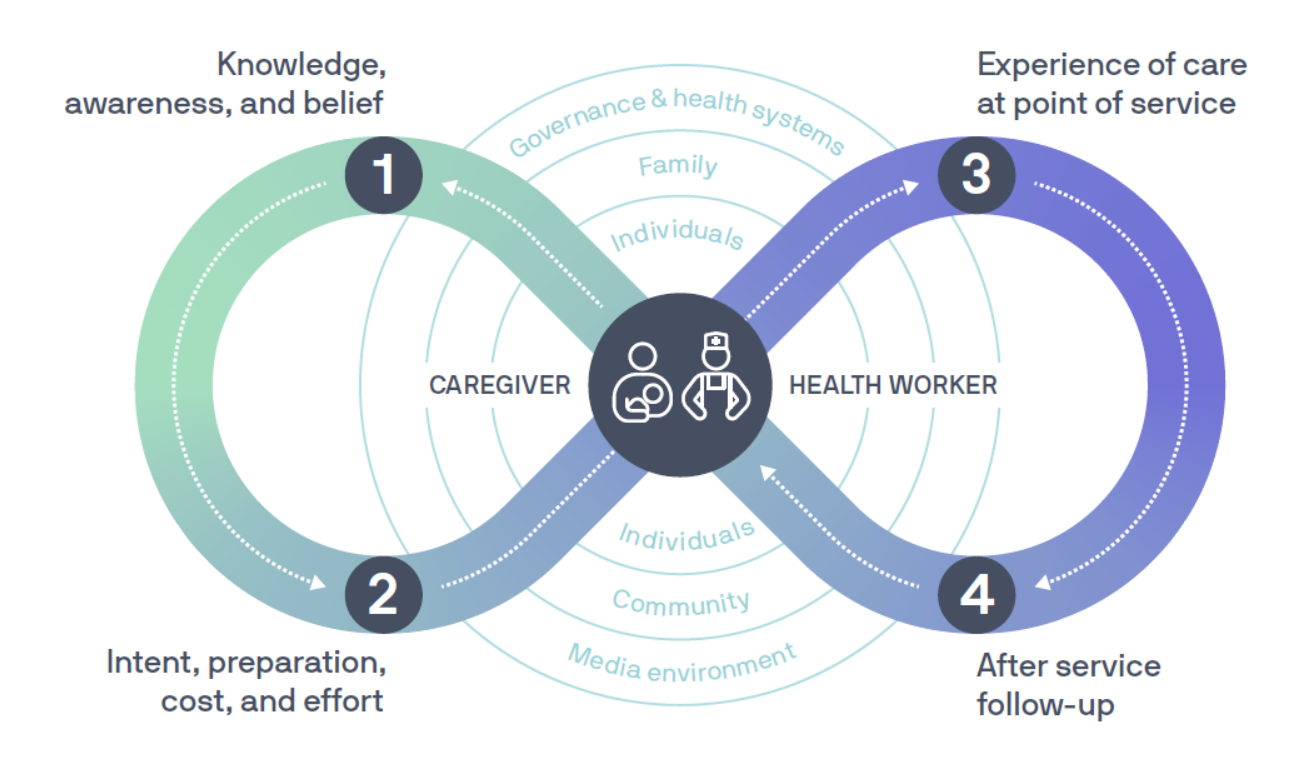
Adapted from UNICEF Health Section Implementation Research and Delivery Science Unit and the Office of Innovation Global Innovation Centre. Demand for Health Services Field Guide: A Human-Centred Approach. UNICEF; 2018.
Product and service delivery adaptations
Stakeholders identified product and service delivery adaptations—such as using one‑dose vials and flexible cold chain approaches—as key to improving timely hepatitis B birth‑dose delivery, especially outside facilities. One‑dose vials were preferred for efficiency and reduced wastage, while controlled temperature chain (CTC) options offer practical solutions to address cold chain gaps, power interruptions, and improve ability to reach newborns delivered in out-of-facility settings. However, some stakeholders expressed concerns about added training complexity and costs for CTC-approved vaccines, while manufacturers highlighted the resource-intensive WHO CTC prequalification process as a barrier.
Human-centered design to co-create implementation strategies
PATH’s human‑centered design workshops in Ethiopia and Uganda generated community and facility strategies to improve timely birth‑dose delivery, including stronger awareness, early birth notification, better community‑facility communication, and expanded CHW roles. Facility approaches focused on integrating vaccination into delivery workflows and improving access. Countries are now adapting these prototypes to strengthen maternal and newborn health services. For more details, please see the human-centered design summary report linked in the resources section below.
Recommendations and opportunities for continued learning
The project identified recommendations for strengthening health systems, integrating birth‑dose vaccination into maternity care, using both one‑ and ten‑dose vials strategically depending on delivery setting, and expanding CHW roles, including possible home‑based vaccination. Countries should locally adapt implementation strategies for increasing birth-dose coverage and tracking timeliness, while global partners refine demand forecasts, streamline CTC prequalification, expand support for one‑dose procurement in community settings and low-volume facilities, and promote cross‑country learning.
Further learning should assess how human‑centered design strategies perform in real world settings, including rapid home‑birth identification, CHW task‑shifting, integration with early postnatal services, and private sector engagement to ensure birth-dose administration in private facilities. Research is also needed on product adaptations including use of mixed (one- and ten-dose) vaccine presentations, quantifying settings where CTC adds the most value, and evaluating CTC-approved vaccines’ impact on timely coverage. Future studies should also explore the feasibility and acceptability of microarray patch administration by CHWs.
This synthesis brief identifies learnings across the four project countries, including barriers and facilitators, product and service delivery adaptations, and recommendations for hepatitis B birth-dose delivery within and outside of facility settings.
These four in-depth case studies outline each country’s context, current strategies, and study sites and design, as well as barriers, facilitators, and recommended service delivery strategies to ensure timely hepatitis B birth-dose delivery.
These briefs summarize the case studies in each project country, highlighting policy and program strategies for increasing timely hepatitis B birth-dose coverage.
This report summarizes learnings from human-centered design co-creation workshops held in Ethiopia and Uganda to generate community and facility-based strategies for timely hepatitis B birth-dose delivery.
During this March 2026 webinar, a diverse panel of experts shared evidence and insights from Ethiopia, The Gambia, Nigeria, and Uganda to evaluate the feasibility, acceptability, cost, and impact of strategies to improve timely hepatitis B birth-dose vaccination for infants born in both facility and community settings.
Header photo: PATH/Khalid Alli


