As more countries begin rolling out COVID-19 vaccines, many people are asking the same question: When will life return to “normal”?
The answer depends on a range of complex factors, from vaccine supply and distribution, to willingness to receive the vaccine, to timely delivery of the second dose. But ultimately, a return to “normal” relies on one factor above all others: herd immunity.
A well-known concept among epidemiologists, herd immunity became a household term when the COVID-19 pandemic began last year. Herd immunity refers to the protection from an infectious disease that occurs when a sufficient proportion of a population can no longer acquire or transmit infection, either through vaccination or immunity resulting from previous infection.
Through herd immunity, the virus struggles to find susceptible hosts, and the chains of transmission break down, so even those who aren’t immune (for example, people who can’t receive a vaccine for health reasons) are protected.
Herd immunity rarely occurs through natural disease spread alone, and it rarely brings an end to an epidemic. Most experts are certain it won’t end the COVID-19 pandemic and have highly advised against herd immunity as a response strategy, warning of the high morbidity and mortality that would result.
However, now that vaccination has begun, herd immunity could be within reach. The catch? We don’t really know when it will happen.
Dr. Anthony Marfin, medical epidemiologist in Vaccine Impact Research at PATH’s Center for Vaccine Innovation and Access, says understanding the determinants of herd immunity can explain why.
Herd immunity is a moving target
Herd immunity is partly dependent on a virus’ basic reproduction number, R0 (pronounced “R naught”), a measure of contagiousness or transmissibility. R0 represents the number of secondary infections resulting when one infection is introduced into a completely susceptible population.
R0 can be highly variable for any given virus, but in general, an R0 greater than 1 means infections will increase over time and an R0 less than 1 means infections will decrease.
Scientists and epidemiologists often use R0 to help predict the threshold of immunity that is needed to stop infection transmission within a population. Although it is an oversimplification, a higher R0 means a higher proportion of a population must be immune to stop transmission. For example, the R0 for measles is estimated to be about 12 to 18.
“This means that when the measles virus first enters a population where nobody has immunity, one infected person will result in 12 to 18 other infected people. That transmission will continue until roughly 95 percent of the population is immune,” says Dr. Marfin. “That 95 percent is the herd immunity threshold.”
Sustaining herd immunity requires constant maintenance. After decades of vaccination, measles was eliminated in the United States in 2000. But in communities that don’t maintain high vaccination coverage, pockets of outbreaks are appearing.
Many public health experts say R0, a number inherently based on assumptions, has been misused and misunderstood throughout the current pandemic. Calculating R0 is far from straightforward, and it can vary greatly from one population to another. For example, different communities follow different non-pharmaceutical interventions—such as physical distancing—to varying extents, influencing the specific R0 for that community.
Furthermore, there is discussion among experts as to whether R0 is even the best number to use for talking about herd immunity. Today, it may be better to use R, the effective reproduction number, which accounts for the lower susceptibility to infection within a population due to vaccination (R0 assumes complete susceptibility)—or Rt, which is R specified at a particular time.
Dr. Marfin says the current predictions for COVID-19’s herd immunity threshold are still just that—predictions—and will be continuously refined as we learn more about the virus.
“Currently, the basic reproduction number for SARS-CoV-2 is estimated between 2 and 3. Based on that, initial estimates put the herd immunity threshold around 60 to 70 percent to stop transmission,” he says. “But we need to remember that R0 is only one part of this estimate. There are many reasons why some estimates may be higher.”
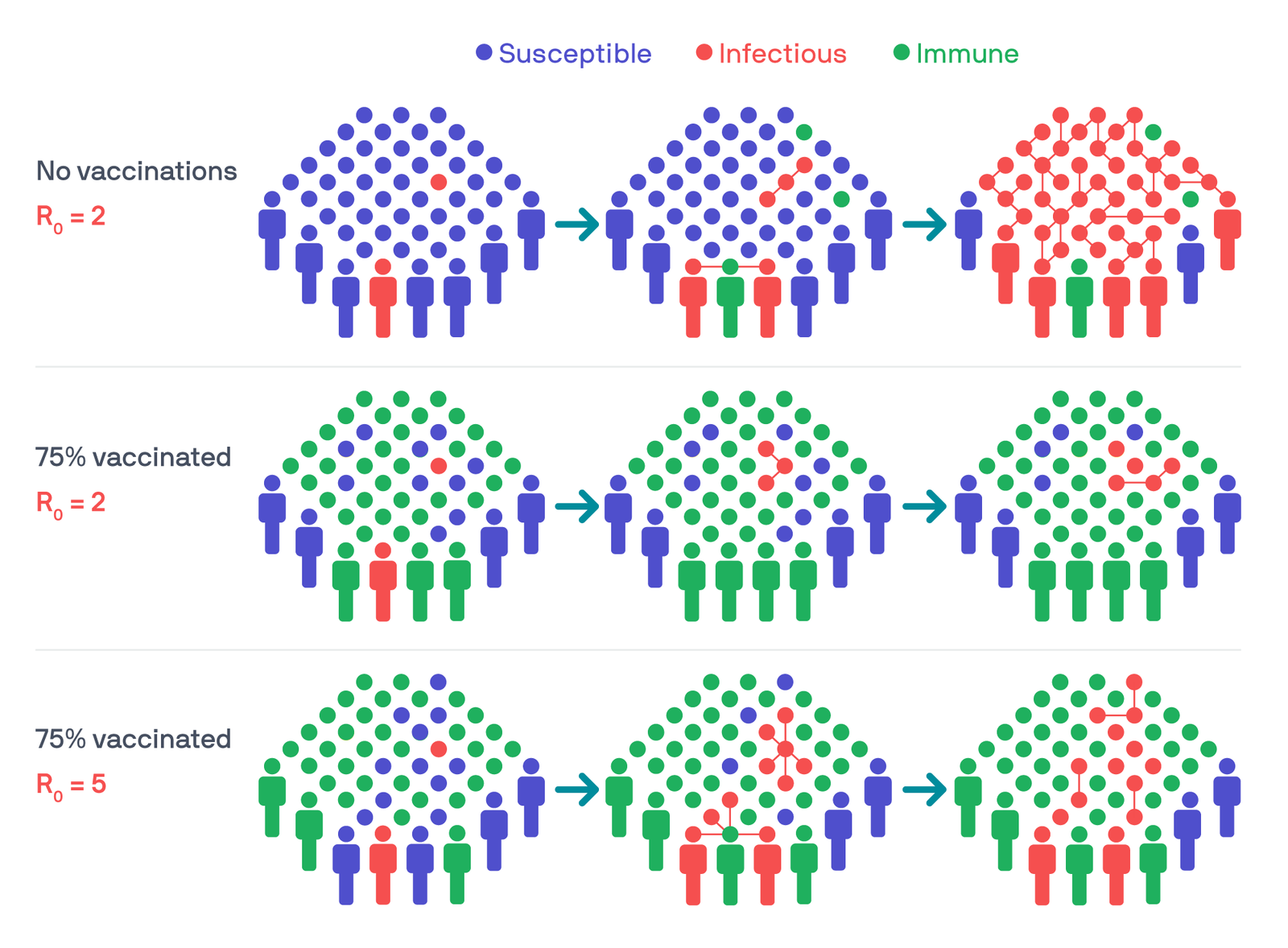
Three example scenarios show how reproduction numbers and immunity levels can affect how a virus spreads through a population. Illustration: PATH/Thom Heileson.
Virus mutations bring more uncertainty
If the virus mutates, the R0 can change—and SARS-CoV-2 is already mutating.
Three notable variants have emerged in Brazil, South Africa, and the United Kingdom—but there could be more. The viral sequencing capacity required to identify new variants is limited in many countries.
“These variants are not surprising,” Dr. Marfin says. “Whether it’s HIV, influenza, West Nile, or SARS-CoV-2 —RNA viruses mutate. It’s what they do.”
New variants might have a different R0. The variant identified in the UK, known as B.1.1.7, spreads more easily and likely has a higher R0.
New variants might also mean antibodies from past variants or vaccination may not protect you as well, or at all. When infected or vaccinated, the body develops antibodies that recognize a particular shape of spike protein on the virus’ surface. But if the shape of the spike significantly changes, our bodies might not recognize it. This would mean the R will increase.
This is the concern with the variant in South Africa, known as B.1.351. Early studies have shown that antibodies from existing vaccines against COVID-19 might not be as effective at attaching themselves to this version of the virus. South Africa stopped using the AstraZeneca-Oxford vaccine after new evidence emerged showing it might not work well against this variant. Of course, vaccine efficacy will have an impact on herd immunity thresholds.
This modeling helps illustrate how: a vaccine with 100 percent efficacy that gives lifelong protection, with an assumed R0 of 2.5 to 3.5, results in a herd immunity threshold around 60 to 72 percent. If vaccine efficacy drops to 80 percent, then the threshold becomes 75 to 90 percent.
And lifelong protection can't be assumed.
Immunity may not be permanent
Immunity against any disease only lasts as long as our bodies retain immune memory—the ability to recognize an antigen and reinitiate an immune response.
Polio vaccinations, for example, protect for many years. This, combined with widespread vaccination coverage, has brought the world close to eradicating the disease. Others, like influenza vaccination, must be administered every year.
A recent study found that natural immunity—from previous infection—to COVID-19 can last at least eight months. It’s still too early to tell how long immunity from vaccination will last. If it isn’t long-lasting, it’s possible that vaccinations against SARS-CoV-2 might be needed annually, like the influenza vaccine.
“We’re going to have to continue to monitor,” Dr. Marfin says. “But in the meantime, keep wearing your masks.”



