While molecular tests historically have been used in well-equipped laboratories with trained technicians, recent technological advances in assay chemistry, cartridge design, instrumentation, and new specimen collection methods are pushing the capacity of molecular testing closer to patients, offering the promise of high‑quality test results on-site.
The COVID-19 pandemic highlighted the critical role of accessible, reliable diagnostics in detecting and responding to outbreaks at every level of the health system—and this unmet need accelerated the development of new molecular products appropriate for use at or near the point of care (POC). Examples of these technologies include low-complexity automated nucleic acid amplification tests (LC-aNAAT) and near POC NAAT for tuberculosis.
However, technological innovation alone does not guarantee real-world health impact: New tools must be compatible with the people, workflows, and environments where they are used to be impactful.
Why usability matters
Understanding and incorporating the user experience—how and where a test is run and what challenges may emerge—is essential for creating impact through effective adoption in health systems. For instance, ongoing efforts by TB and HIV programs to achieve onsite impact with decentralized molecular diagnostics demonstrate that effective technology is not enough: strong training, quality assurance, maintenance, and supply chains, as well as clear linkages to treatment, are critical factors.
PATH has long championed the use of human-centered design throughout all phases of the product development lifecycle. Prioritizing user feedback helps to ensure innovations are appropriate for both the intended users and their context of use.
“User insights allow us to identify usability issues while they are actionable, translate critical needs into product requirements, or refine prototypes to help diagnostic developers introduce products optimized for success.”— Emily Gerth-Guyette, Advanced R&D Officer, Diagnostics
Emerging molecular diagnostic technologies
Grounded in these principles, PATH conducted usability evaluations of six near-POC molecular technologies, beginning with expert assessments in a controlled laboratory setting and extending to end‑user evaluations in Brazil and Mozambique.
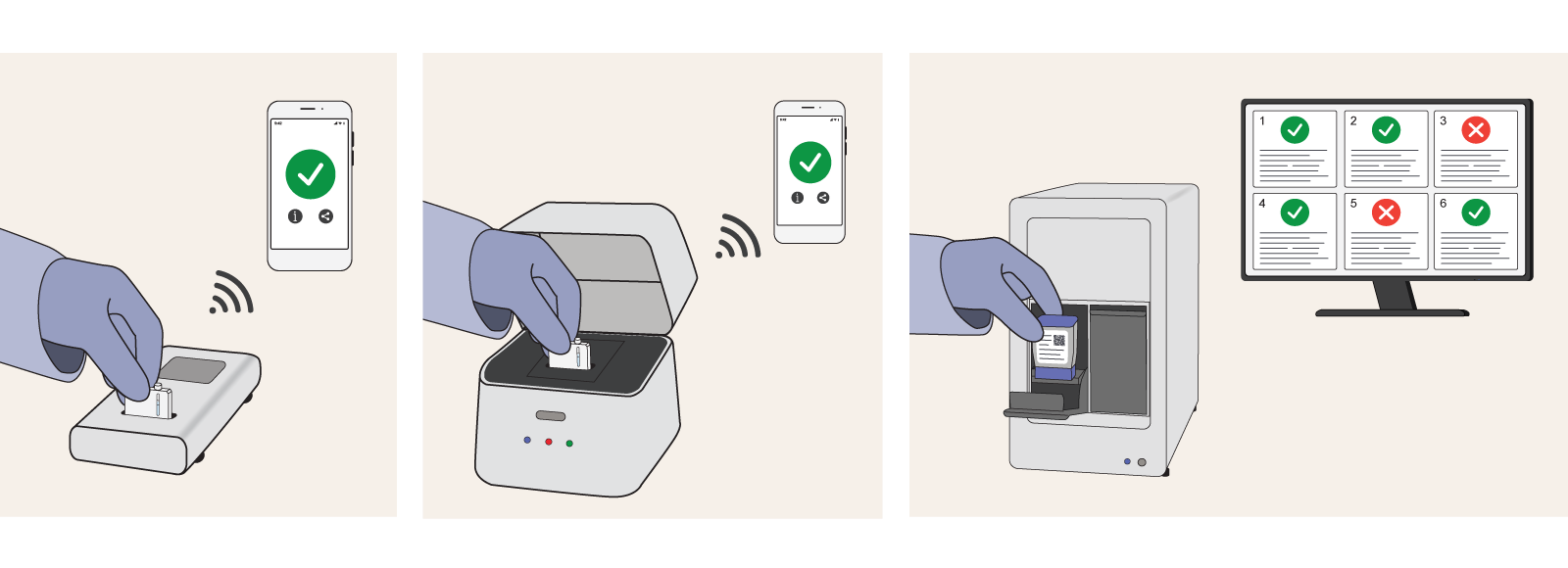
Examples of molecular diagnostic platforms that are similar in function but range in format from compact, handheld devices to larger technologies the size of a desktop computer. Image: PATH/Patrick McKern.
Although the six platforms differed slightly in size and format, all were indicated for the detection of respiratory infections (including SARS-CoV-2, influenza, and RSV); used single-use, disposable test cartridges; and processed one specimen at a time.
While functionally similar, workflows and instrument formats varied by product. For instance, two platforms were integrated with mobile applications to guide users through workflows and display results. All assays were indicated for use with nasal and/or throat swabs, though some were also compatible with saliva or oral rinse specimens.
We conducted in‑house assessments of five technologies with PATH product design and development experts. In each test, seven to nine participants completed the workflow using written instructions and simulated samples and insights were captured through structured observations and questionnaires. Expert users with experience in analytical laboratory testing of human samples also completed a heuristic‑based usability assessment to identify design strengths and areas for refinement.
End-user evaluations in Brazil and Mozambique
To incorporate end‑user perspectives, a sixth product was evaluated in Brazil and Mozambique. PATH collaborated with public health reference laboratory Laboratório Central de Saúde Pública (LACEN) in Brazil and with researchers at the Eduardo Mondlane University Medicine College (FAMED-UEM) in Mozambique. In both countries, we received feedback informed by stakeholder interviews with local laboratory and public health experts in combination with moderated, hands-on usability sessions with health care workers who performed the test.

A simulated demonstration of diagnostic platform features during a usability evaluation in Brazil. Photo: PATH/Mikka Nyarko.
Four key insights
Across our evaluations, we gleaned four insights key to usability and implementation considerations for emerging molecular diagnostic innovations.
1. Most products demonstrated strong usability. Products with clear instructions for use and an intuitive workflow were easy for participants to use. Conversely, if instructions for use were unclear or if the workflow was not intuitive, we observed more steps with a high risk of failure—highlighting the importance of well-sequenced processes and product design choices that minimize ambiguity.
2. Key product characteristics determine implementation fit.
- Portability and power requirements were cited as especially important for community‑based and primary care environments, where infrastructure may be limited.
- Cost—both for instruments and consumables—strongly influenced perceptions of long‑term sustainability.
- Sample preparation complexity plays a key role in determining if tests can be operated by users without laboratory training.
- Throughput levels, or the volume of results generated at a time, can limit utility. Diagnostic needs currently met by high-volume testing platforms will be difficult to address with some of the low-throughput POC technologies included in the assessment. For instance, to be effective for surveillance, it must be feasible for platforms to batch test.
- Turnaround time to results is a critical factor. To be effective at the point of care results must be delivered quickly and directly inform clinical decisions or actions that improve patient outcomes.
Combined, these key characteristics shape how—and where—technologies can be most effectively deployed.
3. Use cases must be clearly defined. This is critical for determining where decentralized molecular testing can be impactful. Evaluators generally found these technologies appropriate for use in resource-constrained locations, but only when matched to the right context and setting and when there is a clear opportunity to address a clinical need. Identifying appropriate use cases requires considering factors such as specimen type, diagnostic alternatives, turnaround‑time expectations, market size, and the associated treatment pathway.
4. Cost-effectiveness is linked to context. There is a critical need to establish where near‑patient molecular testing is—and is not—cost‑effective to guide investment, procurement, and scale‑up. Value is highly dependent on the specific use case and how test results influence clinical decision‑making, patient outcomes, and downstream resource use.
“A quick diagnosis helps us decide on how and where the patient should be treated immediately, which reduces the time and costs of transferring. A quick result is very important.”— User feedback, Brazil
Cost-effectiveness encompasses instrument, maintenance, and consumable costs alongside any savings from faster or more accurate diagnosis, improved treatment, or reduced follow‑up visits. For example, for SARS-CoV-2, this means clarifying where molecular testing at or near the POC can provide meaningful advantages over lower‑cost lateral flow rapid tests or higher‑throughput centralized molecular platforms.
Looking ahead: Decentralized multiplex molecular diagnostics
Findings from these evaluations reinforce that the success of decentralized molecular diagnostics depends on usability, product fit, and comparative value—echoing learnings from global efforts to introduce POC molecular testing for COVID-19, TB, and HIV.
As an expanding pipeline of new products is prepared for market introduction, there is an even greater need for rigorous usability assessments to ensure these technologies can be effectively adopted and used in real-world settings. Moreover, by carrying forward these valuable lessons, we can create the enabling conditions needed for molecular diagnostics to have a positive clinical impact.
Looking ahead, decentralized molecular diagnostics could facilitate multiplexing or integrated testing—the ability to detect multiple pathogens from a single sample. Many platforms have the technical capacity to detect not only respiratory infections but also febrile illness, sexually transmitted infections, and/or antimicrobial or drug resistance markers. Especially in clinical settings where symptoms may have many possible causes, multiplex tests have the potential to add significant value.

Staff in PATH’s product development laboratory review test results. Photo: PATH.
Realizing the potential of multiplexing technology will require a careful balance, considering clinical utility and cost while ensuring test results can be integrated into existing treatment protocols and health information systems.
Ultimately, user-centered product development helps ensure that molecular diagnostics do more than reach the point of care—they deliver real benefits for communities.



